Exercise Boosts Nerve Repair: AMPK Pathway Enhances Axon Regeneration
Research Summary: Physical exercise engages AMPK signalling in body wall muscle and neuron to enhance axon regeneration, leading to better functional recovery. AMPK activation through AICAR improves axon regeneration.
Researcher Spotlight

Dr Sibaram Behera is a Research Associate at BRIC-National Brain Research Centre, Manesar.
Linkedin | Twitter | Instagram
Lab: Dr Anindya Ghosh Roy, BRIC-National Brain Research Centre, Manesar
Lab twitter: https://x.com/GhoshRoyLab
Lab website: https://anindyaghoshroy.weebly.com/
What was the core problem you aimed to solve with this research?
Injury to the nervous system disrupts neural circuits, resulting in behavioural impairment. Neural circuit repair in humans is inefficient due to limited axon regeneration capability. Thus, Injury to the central nervous system is a global socio-economic challenge, and currently, there is no effective cure. Research efforts from the past decades have improved our understanding of the axon regeneration mechanism in the mammalian nervous system, revealing many cell-intrinsic and extrinsic factors. The ongoing research in the field is to manipulate these factors in combination to improve the functional recovery. Recent findings highlight promising outcomes of rehabilitative exercise training on improving functional recovery as a stand-alone paradigm or in combination with other modalities. However, a clear understanding of the effect of exercise on axon regeneration is not well elucidated. We need to understand the underlying mechanism through which exercise improves axon regeneration and recovery to efficiently utilise it as therapy. Also, finding novel exercise mimics will help to design better therapeutic strategies.
Therefore, our core objective was to understand the molecular mechanism of the effect of physical exercise on axon regeneration.
“Physical exercise promotes nervous system repair through activation of cellular energy-sensing pathways and represents a promising therapeutic avenue” –Dr Anindya Ghosh Roy
How did you go about solving this problem?
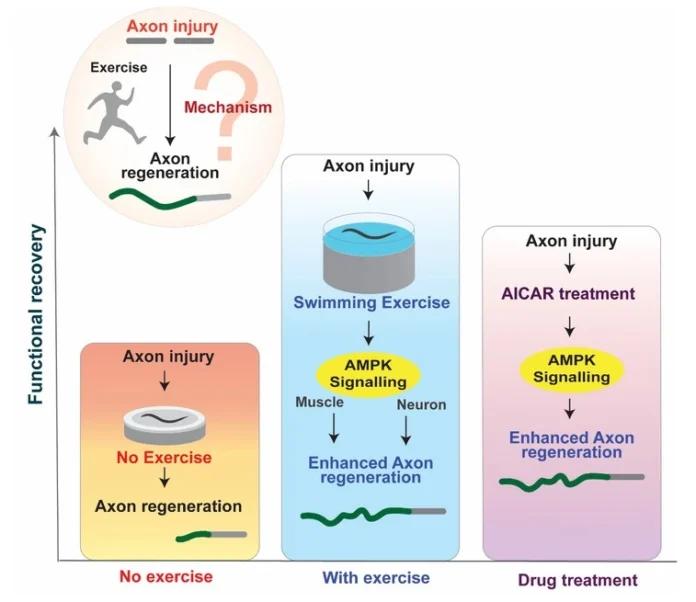
Our approach was to use a simple system to understand the underlying molecular mechanism of exercise’s effect on axon regeneration. In this direction, in our previous work, we combined the PLM mechanosensory neuron axon injury model of Caenorhabditis elegans with swimming exercise to establish a simple and efficient paradigm showing exercise’s axon regeneration-promoting effects. We found a lead that the metabolic sensor, AMPK, is an important mediator of exercise’s positive effects. Working on this lead, we further used the paradigm to dive deeper into understanding the downstream signalling mechanism of exercise. First, we characterised the role of AMPK signalling through genetic analysis, imaging experiments and pharmacological treatments, then characterised the upstream and downstream molecules of AMPK. Exercise induces a systemic effect by affecting multiple tissues, and AMPK also works in multiple tissues. Therefore, one of our focuses was to understand the tissue-specific role of AMPK signalling in the context of exercise-mediated axon regeneration enhancement. In this direction, we extensively characterised axon regeneration outcomes in tissue-specific knockdown conditions. Also, we have strengthened the finding through fluorescence reporter analysis. We also tested pharmacological targeting of AMPK as a potential strategy for improvement of functional recovery after an injury. Overall, we implemented multiple experimental analyses to understand the role of AMPK signalling in exercise-mediated axon regeneration.
How would you explain your research outcomes (Key findings) to the non-scientific community?
We wanted to understand whether physical activity can help damaged nerve cells recover. Using a tiny worm often used in research, we developed a simple way to study how exercise affects injured nerves. We found that even a single session of activity can speed up the repair of damaged nerve fibres (axons), helping restore lost functions more effectively.
Digging deeper, we discovered that this benefit is driven by a key internal “energy-sensing” system in cells, known as AMPK. This system acts like a central switch that turns on repair processes in both muscles and nerve cells. Several important helper molecules work alongside AMPK to carry out this repair program.
Excitingly, we also showed that a drug that activates this same system can mimic some of the positive effects of exercise. This suggests that, in the future, it may be possible to develop treatments that promote nerve repair by targeting the same pathways triggered by physical activity.
What are the potential implications of your findings for the field and society?
Our findings highlight the powerful role that physical activity can play in helping the nervous system recover after injury. By identifying AMPK as a key molecular link between exercise and nerve repair, this study opens new directions for developing therapies that could mimic the benefits of exercise, especially for individuals who are unable to engage in physical activity due to injury or illness. Targeting this pathway with drugs may offer a promising strategy to enhance nerve regeneration and improve functional recovery.
For the scientific field, these results underscore the need to better understand and optimise “exercise-based” interventions as part of treatment strategies for nerve damage. They also point to the importance of studying how different tissues in the body work together to support recovery.
What was the exciting moment during your research?
One of the exciting moments was when I tested the mutants of daf-16 and mdt-15 for exercise’s effect and found that the enhanced phenotype was absent in these mutants. This was exciting in the context of the whole project and motivated me to further characterise the complex signalling. Another exciting moment in the whole project was seeing exercise like phenotype with just AICAR treatment in the absence of a swimming exercise session. Such moments kept me motivated to overcome the challenges associated with the study.
Paper reference: Behera S, Ghosh Roy A (2026) Exercise-induced differential transcriptional output of AMPK signalling improves axon regeneration and functional recovery. J Neurosci.46 (9) e0124252026. https://www.jneurosci.org/content/46/9/e012425202.
Explore more
🎤 Career – Real career stories and job profiles of life science professionals. Discover current opportunities for students and researchers.
💼 Jobs – The latest job openings and internship alerts across academia and industry.
🛠️ Services – Regulatory support, patent filing assistance, and career consulting services.