New Breakthrough in Parkinson’s Research: How Neuronal Membranes Drive Protein Aggregation
Research Summary: We uncovered a previously unknown lipid membrane surface-mediated mechanism driving α-Synuclein (α-Syn) aggregation in the neurons. The membrane surface acts as a key regulator of α-Syn condensate formation and fibril nucleation.
Researcher Spotlight

Jafarulla Shaikh is a Ph.D. scholar in the Membrane Biology Lab at the School of Biological Sciences, NISER. He earned his B.Sc. and M.Sc. in Biochemistry from the Aligarh Muslim University. His research focuses on dissecting the interplay between membrane properties and α-Synuclein aggregation in Parkinson’s disease pathogenesis using in vitro reconstitution and cell biology.
LinkedIn: https://www.linkedin.com/in/jafarulla-shaikh786/
Twitter: https://x.com/jskrockstar
Instagram: https://www.instagram.com/jafthepunisher
Lab: Dr. Mohammed Saleem, National Institute of Science Education & Research (NISER), Bhubaneswar
Lab website: https://www.saleemlab.co.in/
What was the core problem you aimed to solve with this research?
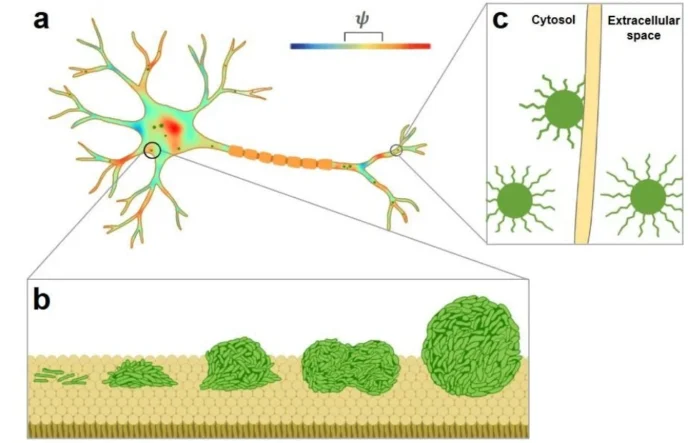
We aimed to understand how α-Syn aggregates inside neurons at physiologically relevant concentrations. α-Syn, a small neuronal protein abundantly expressed in the brain, has long been implicated in the pathogenesis of synucleinopathies due to its propensity to misfold and aggregate into amyloid fibrils. These fibrillar deposits are hallmarks of Parkinson’s disease, but the precise cellular triggers that initiate α-Synuclein aggregation have remained elusive. Previous studies have shown that α-Syn undergoes phase separation only at artificially elevated concentrations, well beyond what neurons actually contain, raising the question of whether such high levels are truly required to form aggregation-competent species that seed toxic fibrils in patients. Additionally, the role of membranes in this process remains poorly understood. Using advanced confocal microscopy, cell biology, and biophysical approaches, we demonstrate that membrane stoichiometry governs the membrane interfacial potential, which drives α-Syn condensation via liquid-liquid phase separation. The resulting condensates serve as nucleation hubs for amyloid fibril growth, ultimately leading to membrane deformation and rupture.
How did you go about solving this problem?
How membrane surfaces regulate the phase separation dynamics of α-Synuclein is still not well defined. To address the above question, we began by reconstituting purified α-Syn on supported lipid bilayers (SLBs) mimicking neuronal membranes to establish a binding platform. Using thioflavin-T assays with membrane vesicles, we observed that fibrillation initiated around 20 hours, followed by slow elongation and a plateau phase. Long-duration imaging of labeled α-Syn on SLBs revealed that condensation occurred via liquid-liquid phase separation (LLPS) without molecular crowding over 24 hours. Surface condensation required as little as 10 nM α-Syn on neuronal membrane mimics over 27 hours, with higher concentrations increasing condensation intensity, indicating sensitivity to lipid composition. Fluorescence recovery after photobleaching (FRAP) showed that mature condensates recovered more slowly than nascent ones, reflecting a liquid-to-solid transition. Biophysical analyses linked condensate formation to reduced membrane tension and compressibility, governed by a narrow interfacial potential range of phosphatidylserines. Primary neuron experiments demonstrated that depolarization promotes α-Syn condensation, membrane association, and extracellular puncta release, facilitating neuron-to-neuron transmission in Parkinson’s disease pathology.
“Membrane’s electrostatic landscape offers early therapeutic targets to stabilize potential and stop Parkinson’s onset.” – Dr. Mohammed Saleem
How would you explain your research outcomes (Key findings) to the non-scientific community?
Imagine your brain cells have walls-called a membrane, just like a house. We discovered that these walls have an electrical charge, which acts like a magnet for a protein called α-Syn. When the charge is just right, it pulls the protein out of the fluid inside the cell and causes it to clump together on the wall’s surface, even when the protein is present in tiny, perfectly normal amounts. These clumps then grow into the rigid, hair-like structures that physically damage and break the membrane. Alternatively, brain cells’ own walls, depending on their electrical charge, can inadvertently trigger the very clumping process that leads to Parkinson’s disease, and your brain’s natural activity can then spread that damage to the neighbouring brain cells. In short, the brain cells’ own wall and its electrical activity may be key drivers of how Parkinson’s disease starts and spreads- a finding that opens new exciting doors for the treatment.
What are the potential implications of your findings for the field and society?
This research fundamentally reframes Parkinson’s disease, not just a protein problem, but as a membrane physics problem. This work represents a seminal advance in our understanding of neurodegeneration, highlighting how the electrochemical landscape of membrane interfaces acts as a fundamental driver of α-Syn pathology. By uncovering the intricate interplay between membrane biophysics and aggregation, our findings not only shed light on the underlying mechanisms of disease but also point toward innovative therapeutic strategies-ones that focus on modulating the cellular microenvironment rather than targeting proteins alone.
What was the exciting moment during your research?
One of the most exciting moments in the research was likely when we observed how α-Syn interacts with the neuronal membrane mimic and leads to the liquid condensate formation in physiologically relevant concentrations, without the molecular crowder breaking the traditional view of condensate formation in very high concentrations of protein with molecular crowder. Another astonishing finding reveals a paradigm shift by highlighting the supremacy of the membrane interface physicochemical properties. This interfacial potential serves as a potential governor for the phase transition of α-Syn from monomeric state to condensed oligomers on the neuronal membrane surface, which leads to the fibrillation. This fibrillation leads to membrane deformation and damage, directly witnessing how the membrane interface regulates the α-Syn aggregation mechanism.
Paper reference: Shaikh, J., Nagarajan, A., Mitra, T. et al. Membrane interfacial potential governs surface condensation and fibrillation of α-Synuclein in neurons. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70840-2
Explore more
🎤 Career – Real career stories and job profiles of life science professionals. Discover current opportunities for students and researchers.
💼 Jobs – The latest job openings and internship alerts across academia and industry.
🛠️ Services – Regulatory support, patent filing assistance, and career consulting services.