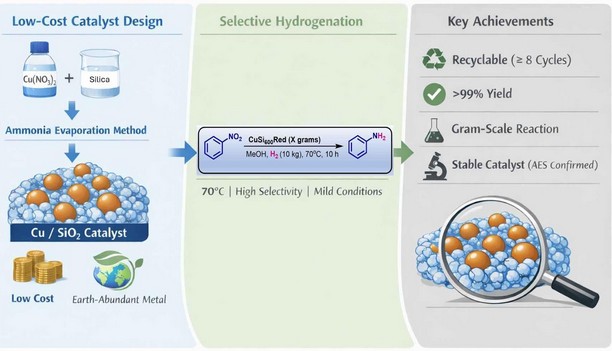
Research Summary: We developed a recyclable copper-silica catalyst that efficiently converts nitroarenes to anilines under mild conditions with >99% yield and excellent selectivity, offering a sustainable alternative to expensive noble-metal catalysts.
Researcher Spotlight

Anjali Ankush Katkar is a catalysis researcher working on sustainable heterogeneous catalysts, focusing on earth-abundant metal systems for efficient hydrogenation and environmentally responsible chemical synthesis.
Linkedin: linkedin.com/in/anjali-katkar-649b13132
Instagram: Katkar_anjali
Lab: Dr. Anant R. Kapdi, Kapdi lab – Chemistry Department, Institute of Chemical Technology, Matunga Mumbai
Website: https://kapdigroupresearch.com
What was the core problem you aimed to solve with this research?
Selective hydrogenation of nitroarenes to anilines is a cornerstone transformation in the chemical industry, yet it often relies on precious-metal catalysts such as palladium or platinum, which are expensive and limited in supply. Many existing catalysts also suffer from poor recyclability or harsh reaction conditions. Our goal was to develop a cost-effective, earth-abundant, and recyclable catalyst that could achieve high efficiency and selectivity under mild and practical conditions while reducing dependence on noble metals.
How did you go about solving this problem?
We designed a silica-supported copper catalyst using a controlled ammonia-evaporation deposition strategy, employing readily available and inexpensive sources of both silica and copper, making the approach economically attractive and scalable. This method enabled uniform dispersion of active copper species within the silicate matrix. Through extensive characterization including X-ray diffraction, electron microscopy, surface spectroscopy, and reduction studies, we established how the catalyst structure governs its activity. This systematic understanding allowed us to optimize the material to achieve exceptional catalytic efficiency, stability, and recyclability in hydrogenation reactions while maintaining a cost-effective and sustainable catalyst design.
How would you explain your research outcomes (Key findings) to the non-scientific community?
Many everyday products including medicines, dyes, and agricultural chemicals require special chemical reactions to make their key ingredients. We developed a new reusable catalyst made from copper, a widely available metal, that performs one of these important reactions extremely efficiently. It produces the desired chemical with very high purity and yield, works under gentle conditions, and can be reused multiple times, making the process more sustainable and economical.
“As a part of sustainable approach to catalysis, choosing Cu over pd as an earth abundant metal has many advantages if the reactivity can be fine tuned and a carbon – free approach further enhances its appeal.’ – Dr. Anant R. Kapdi
What are the potential implications of your findings for the field and society?
Our findings demonstrate that earth-abundant metals like copper can rival traditional precious-metal catalysts in efficiency and selectivity. This opens new opportunities for designing cost-effective and environmentally responsible catalytic processes. In the long term, such catalysts could help industries produce pharmaceuticals, agrochemicals, and fine chemicals more sustainably, safely, and economically, contributing to greener chemical manufacturing.
What was the exciting moment during your research?
The most exciting moment came when the catalyst consistently delivered near-quantitative yield and complete selectivity, even after multiple recycling cycles. This performance was further confirmed during a gram-scale reaction, where the catalyst maintained the same high efficiency, demonstrating its practical applicability beyond small milligram laboratory experiments. Equally exciting was the detailed analysis of the spent catalyst after repeated use.
Comprehensive characterization showed that the structural features of the catalyst remained largely intact, and importantly, Auger Electron Spectroscopy (AES) provided quantitative evidence that the copper species were well preserved without significant loss or leaching. Observing such sustained catalytic activity, structural integrity, and quantitative stability from a simple copper-based system was truly rewarding and confirmed the strong potential of this catalyst for scalable and practical applications.
Paper reference: Katkar, A., Pal, K., Garade, A. et al. Recyclable and efficient silica-supported copper hydrogenation catalyst. Commun Chem 9, 63 (2026). https://doi.org/10.1038/s42004-025-01867-7
Interview no 413
Explore more
🎤 Career – Real career stories and job profiles of life science professionals. Discover current opportunities for students and researchers.
💼 Jobs – The latest job openings and internship alerts across academia and industry.
🛠️ Services – Regulatory support, patent filing assistance, and career consulting services.