Water-Gated Chemistry in CYP1A Enzymes Reveals Hidden Mechanism of Drug Metabolism
Research Summary: Our research provides insights into the different structural dynamics of two very similar Cytochrome P450 isoenzymes where one isozyme reacts with the substrate and the other reacts barely at all.
Researcher Spotlight

Vaibhav Bhatt is a Ph.D scholar at Molecular Simulation Lab, Shiv Nadar IoE, Delhi-NCR mentored by Dr. Kshatresh Dutta Dubey. His research focuses on the structure-function-activity nexus governing the fate of a protein or an enzyme.
Linkedin: www.linkedin.com/in/vaibhav-bhatt-70a73936
Lab: Dr. Kshatresh Dutta Dubey, Shiv Nadar Institution of Eminence, Delhi-NCR
Twitter: https://x.com/kshatresh
What was the core problem you aimed to solve with this research?
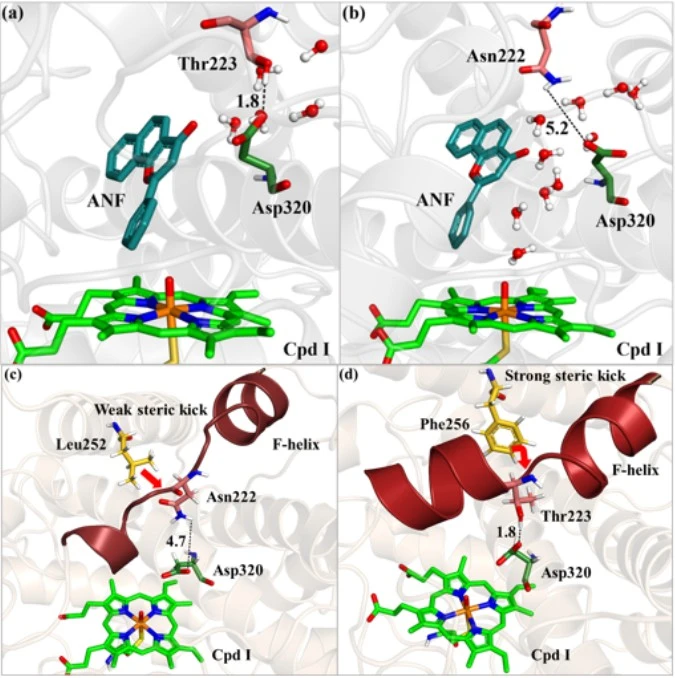
The core problem was to uncover the reason behind differential activity shown by two very similar isozymes. The binding pocket, substrate orientation remains same in both isozymes with similar amino acid residues in the catalytic pocket. Despite this, the enzymes behave differently. One isozyme (CYP1A1) oxidizes the substrate α-naphthoflavone (ANF) while the other (CYP1A2) barely reacts with it. Moreover, the site of oxidation (carbon atom of the substrate where oxygen attacks) was 10 Å away from the oxo-ferryl moiety (source of oxygen for oxidation in CYP450s). So, how could an efficient reaction take place in CYP1A1 if the two atoms that must react are so far apart?
How did you go about solving this problem?
We performed Molecular Dynamics simulations and observed that there are specific residues at the boundary of the catalytic pocket that are different in both the isozymes. Also, since the two atoms that must react together are 10 Å apart, the only other thing in the enzyme-substrate complex that could help in the reaction must be water molecules. Our simulations revealed that in CYP1A1 (active isozyme), one pair of residues, viz., Asn222 and Asp320 forms a water gate that is open and allows water entry into the active site and thereby leading to a successful reaction. On the other hand, in CYP1A2 (non-reactive isozyme) there is the presence of Thr223 in place of Asn222 and the new pair of Thr223 and Asp320 forms a closed gate that disallows water entry into the active site and the reaction does not take place. We further uncovered as to why the two There was a residue (Leu) just behind the Asn in CYP1A1 and a different residue (Phe) across Thr in CYP1A2. Both the residues exerted different steric push to the water gate residues. This explained why the two pairs showed open and closed states. At last, we elucidated the reaction mechanism of this unusual oxidation by CYP1A1 using hybrid QM/MM calculations.
How would you explain your research outcomes (Key findings) to the non-scientific community?
Consider the two similar detoxifying cytochrome P450 enzymes as two washing machines of the same company but one efficiently cleans and the other does not. Our research found that water is the most essential component of this cleaning/detoxification process. Now there must be an inlet from where water enters the machine. But a gate controls the inlet. In the working machine, the gate is open so water flows in. In the inefficient washing machine, the gate is closed so almost no water enters for the cleaning process. We found the reason as to why the gate is open in one machine and closed in the other. We also found exactly how the process of cleaning takes place so difficult dirt can be more easily removed.
Our team has shown how the human body’s detoxification enzymes (Cytochrome P450s) perform a reaction with foreign, many times harmful substances that enter our body through various means and help in their elimination from the human body. We visualize the protein/enzyme world at atomic level to understand why some changes happen and others don’t.
This work shows how minute structural motions and water organization can redefine enzyme function, reminding us that small molecular details govern major biological outcomes. — Dr. Kshatresh Dutta Dubey
What are the potential implications of your findings for the field and society?
By knowing how specific individual units comprising the enzyme move and regulate the enzyme’s function, we can fine-tune the enzyme’s structure. This helps predict drug behavior and design better medicines inspired by nature’s own chemistry. Furthermore, efficient variants of the enzymes can be bioengineered with better drug delivery mechanisms using modern biotechnological tools. So, yes, it is an exciting area of research where we get to understand nature’s machinery from a fundamental, molecular perspective.
What was the exciting moment during your research?
The awe moment for me was to understand the ways of protein’s function by studying the subtle changes in its structure. It is a macro-molecular object but its ability to adopt different conformations gives it an almost living-like status. These conformations are a result of the interplay of amino acids that make up the protein itself. Knowing this fundamental reality of the molecules of life really inspires a philosophical outlook towards the natural sciences.
Paper reference: ACS Catal. 2026, 16, 4, 3988–3998. (https://doi.org/10.1021/acscatal.5c09130)
Interview no 411
Explore more
🎤 Career – Real career stories and job profiles of life science professionals. Discover current opportunities for students and researchers.
💼 Jobs – The latest job openings and internship alerts across academia and industry.
🛠️ Services – Regulatory support, patent filing assistance, and career consulting services.