Dopamine and Rotenone Drive Toxic Protein Changes in Parkinson’s Disease Study
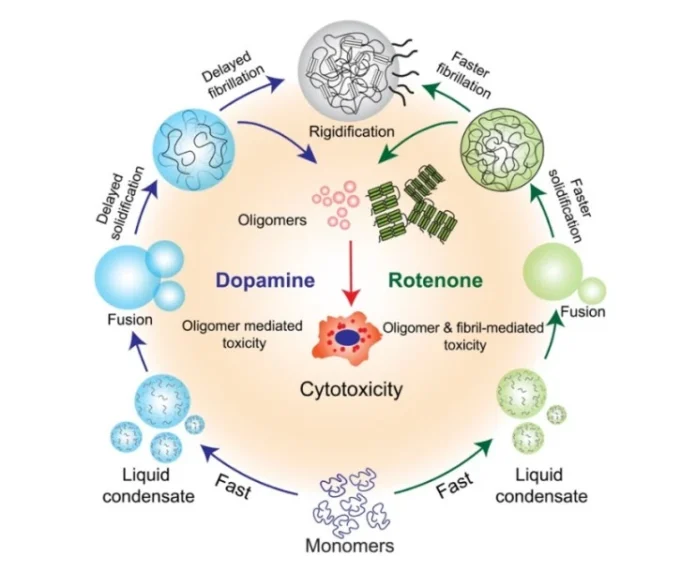
Research Summary: Dopamine and rotenone both accelerate α-synuclein (α-Syn) phase separation, with opposing material transitions: rotenone undergoes rapid solidification, while dopamine remains in a prolonged liquid state; yet both cause cytotoxicity, underscoring a shared pathological convergence in Parkinson’s disease.
Researcher Spotlight

1) Riya Bera is a senior researcher focused on addressing fundamental questions in biomolecular phase separation and its role in neurodegenerative diseases, with a primary emphasis on α-synuclein in Parkinson’s disease. Leveraging her strong foundation in molecular and cellular biology, she investigates the mechanisms of protein phase behavior and its pathological implications.
LinkedIn: https://www.linkedin.com/in/riya-bera-479b72270
2) Shouvik Manna’s work focuses on understanding how α-synuclein undergoes liquid-liquid phase separation (LLPS) to form the pathological aggregates characteristic of Parkinson’s disease. His work investigates how familial mutations and environmental factors influence this process. By identifying the early biophysical triggers of aggregation, he aims to uncover the fundamental molecular mechanisms that drive neurodegenerative disease progression.
LinkedIn: https://www.linkedin.com/in/shouvik-manna-95a37b222
Twitter: https://x.com/Shouvik__Manna
Instagram: https://www.instagram.com/shouvik__manna
3) Ranjit Shaw is also a senior Ph.D. scholar studying biomolecular phase separation and protein aggregation, focusing on α-Synuclein in Parkinson’s disease, integrating molecular biology, biophysics, and translational diagnostics to understand disease mechanisms.
LinkedIn: https://www.linkedin.com/in/ranjit-shaw-947806211
Twitter: https://x.com/ranjitshaw9730
Instagram: https://www.instagram.com/ranjitshaw9730
Lab: Prof. Samir. K. Maji, Protein Engineering and Neurobiology Laboratory, Department of Biosciences and Bioengineering, Indian Institute of Technology Bombay
Lab social media: https://www.bio.iitb.ac.in/~maji/
What was the core problem you aimed to solve with this research?
An overwhelming majority of Parkinson’s cases are sporadic, meaning they arise without a clear genetic link. For decades, researchers have looked for the possible triggers of the disease, including dopamine, which is a neurotransmitter lost in the disease, and rotenone, which is very well documented to be a risk factor for the disease. Yet the precise mechanisms through which they disrupt α-Syn biology at the earliest stages remained poorly understood. Our goal was to understand the earliest molecular events associated with protein aggregation and to determine whether these two agents act through a shared or distinct mechanistic pathway. It felt quite exhilarating and scientifically relevant to establish that connection.
How did you go about solving this problem?
We approached this systematically by combining rigorous in vitro studies with relevant cell-based experiments. Using purified α-Syn, we monitored in real time how dopamine and rotenone influence the formation and material properties of protein condensates using fluorescence microscopy. To confirm our observations in relevance to the disease, we validated the key findings in SH-SY5Y neuroblastoma cells, a well-established neuronal model for Parkinson’s research. Later, we linked the observed phase-separation dynamics to subsequent aggregation outcomes using Thioflavin T fluorescence kinetics and transmission electron microscopy, and examined cellular responses via cytotoxicity assays. This multi-pronged approach allowed us to conclude the effects of these two modulators on α-Syn phase separation.
Our study reveals how environmental molecules reshape α-Syn phase behavior, offering new mechanistic insights into early events of Parkinson’s disease. – Prof. Samir. K. Maji
How would you explain your research outcomes (Key findings) to the non-scientific community?
Inside our brain cells, a protein called α-Syn normally behaves like a liquid and can form tiny droplets, much like oil droplets in water. These droplets are usually harmless and disappear on their own. But in certain diseases, they stop behaving normally and instead turn into sticky clumps that build up and damage brain cells.
We discovered that both dopamine and rotenone accelerate the formation of α-Syn droplets much faster than they would otherwise. However, rotenone rapidly solidifies these droplets, thereby accelerating fibril formation. Dopamine, on the other hand, keeps the droplets in a prolonged liquid state, but that liquid state is still not safe. Both scenarios are highly toxic to neurons, just in different ways. In simple language, whether the brain chemistry goes wrong from within or a toxic pesticide enters from outside, the result is the same. The protein gets trapped in harmful states, leading to a common catastrophic outcome.
What are the potential implications of your findings for the field and society?
Our findings carry implications at various levels. Scientifically, they reposition liquid-liquid phase separation as a critical molecular event in Parkinson’s disease, where both neurochemical and environmental stressors converge, particularly in sporadic disease. This has the potential to reshape how the field frames the beginning of the disease.
Additionally, it has long been held that preventing fibrillar aggregate formation should be the primary therapeutic target for the disease. Our results also provide an additional insight into dopamine-driven condensate formation. Although dopamine is present in several cell types apart from the substantia nigra, dopaminergic neurons are uniquely vulnerable to dopamine-driven toxicity in PD. Despite remaining in a liquid state, they proved significantly toxic. This is most likely because of oligomeric species that form and persist within those condensates. This suggests that targeting the condensate state itself, and not merely end-stage fibrils, may become a therapeutic possibility.
What was the exciting moment during your research?
One of the most exciting moments during this research was when we first observed the distinct effects of dopamine and rotenone on α-Syn condensates under the microscope. Watching the liquid droplets rapidly transition into solid-like aggregates in the presence of rotenone, while dopamine kept them in a dynamic liquid state, was both surprising and fascinating. It was a clear visual confirmation that environmental factors can fundamentally alter the physical behavior of proteins.
Another memorable moment was realizing that these differences were not just structural but also translated into variations in cytotoxicity. That was when the project moved from being an interesting biophysical observation to something with strong biological and disease relevance.
Paper reference: Riya Bera*, Shouvik Manna*, Ranjit Shaw*, Arpita Bhattacharyya, and Samir K. Maji. “Dopamine and Rotenone Modulate α-Synuclein Phase Separation and Liquid to Solid Transition.” Small (2026): e14922. https://doi.org/10.1002/smll.202514922 (*Equal Contribution)
Explore more
🎤 Career – Real career stories and job profiles of life science professionals. Discover current opportunities for students and researchers.
💼 Jobs – The latest job openings and internship alerts across academia and industry.
🛠️ Services – Regulatory support, patent filing assistance, and career consulting services.