Research Summary: Real-time BEST-HSQC NMR monitoring of ubiquitin deamidation by bacterial CIF deamidases enabled precise kinetic quantification, uncovering how closely related enzymes from distinct bacteria differ in catalytic efficiency and substrate modification.
Author interview

Rashmi Agrata is a biochemist interested in ubiquitin biology. Currently a postdoctoral fellow in David Komander’s lab, Australia, she completed her PhD under Ranabir Das at NCBS-TIFR on bacterial deamidation of ubiquitin associated-proteins.
Linkedin: https://www.linkedin.com/in/rashmiagrata/
Twitter: https://x.com/Rashmi_Agrata
Instagram: @rashmiagrata
Lab: Ranabir Das, NCBS-TIFR, Bangalore
Lab social media: https://x.com/RanabirNMR and https://x.com/DrRanabirDasLab
What was the core problem you aimed to solve with this research?
Understanding how bacterial infection alters protein structure at the amino acid level using Nuclear magnetic Resonance (NMR) spectroscopy.
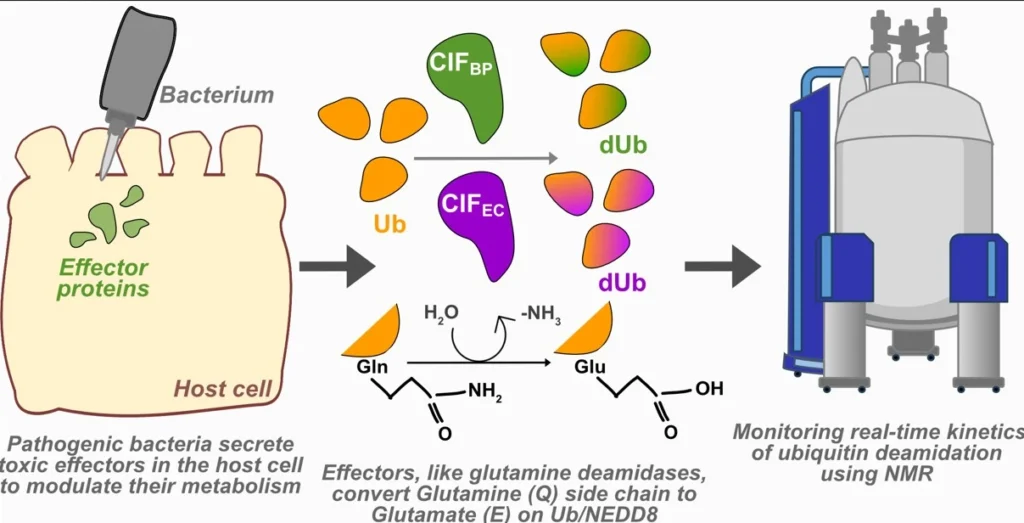
How did you go about solving this problem?
We used NMR spectroscopy to observe how ubiquitin changes during bacterial infection. Since conventional NMR is too slow and sample-dependent to monitor rapid or irreversible protein reactions like deamidation, we employed a faster BEST-HSQC NMR method. By mixing purified bacterial deamidases with ubiquitin in a tube, we tracked spectral shifts in real time, collecting data every few minutes without disturbing the reaction. This setup allowed us to quantify reaction kinetics and directly compare how different bacterial enzymes modify ubiquitin.
How would you explain your research outcomes (Key findings) to the non-scientific community?
Bacteria use powerful enzymes to chemically sabotage the key host protein, ubiquitin. Using a high-tech “camera” (real-time NMR), we showed one enzyme is more efficient than the other. This modification also physically changes distant, crucial parts of ubiquitin, possibly disrupting its function.
What are the potential implications of your findings for the field and society?
We developed a robust, high-resolution kinetic measurement tool with improved accuracy and broad applicability in studying protein modification. The method impacts disease research and drug discoveries. Our study also reveals structural details of ubiquitin modification, opening new avenues to investigate how these changes influence its interactions with other functional proteins.
What was the exciting moment during your research?
The most thrilling moment came when we saw the NMR signals, tiny squiggles on the screen, shifting in real time as the reaction proceeded. That was our “aha!” moment. We suddenly knew that this method could let us watch countless reactions as they happen, almost like having a live broadcast from the molecular world.
Paper reference: Agrata et al, Magnetic Resonance in Chemistry, 2025. https://analyticalsciencejournals.onlinelibrary.wiley.com/doi/10.1002/mrc.70045?af=R
Explore more
🎤 Career – Real career stories and job profiles of life science professionals. Discover current opportunities for students and researchers.
💼 Jobs – The latest job openings and internship alerts across academia and industry.
🛠️ Services – Regulatory support, patent filing assistance, and career consulting services.



