The Shape-Shifter’s Secret: Sugar Metabolism Fuels Fungal Morphogenesis and Infection
Research Summary: Our work elucidates a previously uncharacterized coupling between glycolysis and sulfur metabolism that is critical for driving fungal morphogenesis and virulence, contributing to our understanding of this conserved phenomenon.
Researcher Spotlight

Dhrumi Shah is a PhD student in Dr. Sriram Varahan’s laboratory at CSIR-CCMB, Hyderabad. Her research focuses on elucidating the role of central carbon metabolism during fungal morphogenesis and virulence.
Twitter: @dhrumishah99/@sriram_varahan
Lab: Dr. Sriram Varahan, CSIR-Centre for Cellular and Molecular Biology (CCMB)
Lab website: https://www.sriramvarahanlab.org/
What was the core problem you aimed to solve with this research?
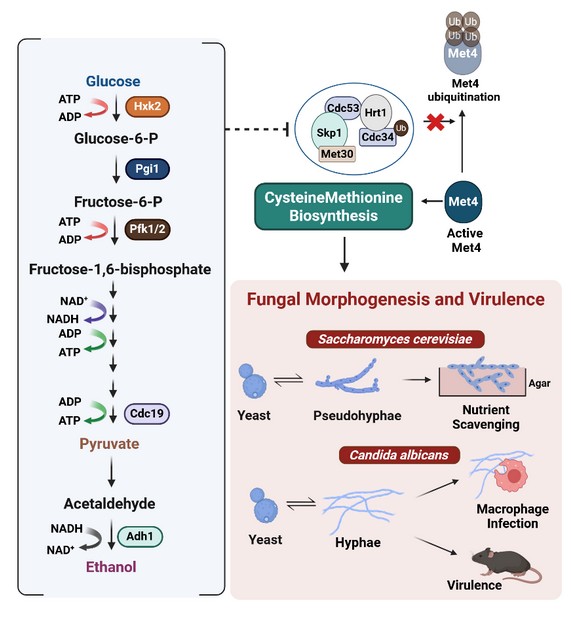
The core problem we addressed was the lack of understanding regarding the metabolic determinants of fungal morphogenesis; while the genetic “blueprints” for fungal morphogenesis were well-mapped, the specific metabolic regulators required for these transitions remained a mystery. We aimed to identify the metabolic triggers that allow fungi to undergo morphogenesis and establish infections. We demonstrated that glycolysis (the primary metabolic pathway that breaks down sugar) is critical for fungal morphogenesis and uncovered a previously unknown coupling between glycolytic flux and the de novo biosynthesis of sulfur-containing amino acids. By identifying this metabolic dependency, we solved the puzzle of how central carbon metabolism acts as a primary signal to drive morphological transitions in fungi which in turn is critical for its virulence within a host.
How did you go about solving this problem?
To address this knowledge gap, we employed a multidisciplinary approach using two fungal species, Saccharomyces cerevisiae and Candida albicans, to systematically map the functional interface between central carbon metabolism and fungal morphogenesis. Our results demonstrate that the perturbation of glycolytic flux causes the downregulation of the de novo biosynthesis of sulfur-containing amino acids, thereby causing the defect in fungal morphogenesis. This metabolic dependency was validated through metabolite add-back experiments, wherein the addition of specific sulfur-containing amino acids successfully bypassed the glycolytic deficit and restored morphological plasticity. Furthermore, using a murine model of systemic candidiasis, we established that the genetic perturbation of glycolysis via the deletion of the gene which encodes for the glycolytic enzyme phosphofructokinase-1 (Pfk1) significantly attenuates fungal survival within the murine host and reduces overall virulence. This virulence defect of the pfk1 deletion strain could be fully rescued when mice were supplemented with exogenous sulfur, confirming that the glycolysis-sulfur axis is a critical determinant of fungal pathogenesis.
Our study, for the first time, demonstrates the regulatory connection between two ancient and highly conserved metabolic pathways (glycolysis and sulfur metabolism) and is an important conceptual advance in understanding fungal morphogenesis. – Dr. Sriram Varahan
How would you explain your research outcomes (Key findings) to the non-scientific community?
Fungi are remarkable biological shape-shifters, transitioning from benign, oval cells (yeast) into aggressive, needle-like structures called hyphae which allows them to penetrate tissues and cause lethal infections. While scientific focus has historically remained on genetic blueprints that orchestrate this shape-shifting ability, new research has uncovered a fundamental “metabolic engine” that drives this transformation. By studying both harmless yeast and dangerous human pathogens, we found that a conserved metabolic axis directly links the rate of sugar breakdown, or glycolysis, to the production of essential sulfur-containing amino acids like cysteine and methionine. When this sugar-processing pathway is obstructed, the fungi become trapped in a harmless state, unable to adopt their invasive form to cause diseases. This internal dependency was confirmed in animal studies, where disabling the fungus’s sugar breakdown pathways left it powerless against the body’s immune cells. Because this discovery targets a core survival mechanism rather than a specialized attack tool of the fungi, it provides a strategic roadmap for developing next-generation treatments that are far more difficult for pathogens to evade or resist.
What are the potential implications of your findings for the field and society?
The identification of the glycolysis-sulfur metabolic axis represents a paradigm shift in fungal pathogenesis, transitioning the focus from transcriptional regulatory networks to the metabolic flux that drives morphogenetic switching. By characterizing the coupling between glycolysis and de novo biosynthesis of sulfur-containing amino acids, this study delineates a critical metabolic requirement for the yeast to hyphae transition. In the context of escalating antimicrobial resistance and the emergence of multi-drug resistant “superbugs”, targeting highly conserved metabolic nodes presents a potent strategy for therapeutic intervention. This approach facilitates the development of precision antifungals designed to attenuate fungal fitness and survival within the host environment. Ultimately, targeting these essential metabolic dependencies offers a robust framework for reducing the morbidity and mortality associated with systemic fungal infections in clinical and immunocompromised settings.
What was the exciting moment during your research?
The most exciting moment of the study was the successful metabolic rescue of the morphogenetic defect with sulfur-containing amino acids. After identifying that glycolysis perturbation resulted in a defect of the yeast to hyphae transition, we discovered that this was due to a downstream failure in sulfur-containing amino acid biosynthesis. The “Eureka” moment occurred when the exogenous supplementation of these specific amino acids, cysteine and methionine, robustly bypassed the glycolytic inhibition and restored the filamentous growth phenotype in both S. cerevisiae and C. albicans and also rescued the virulence ability in C. albicans. Observing this direct functional link between central carbon metabolism and the de novo biosynthesis of sulfur-containing amino acids provided the definitive proof that we had uncovered a fundamental, conserved regulatory axis governing morphological plasticity is fungi.
Paper reference: Shah, D., Rewatkar, N., M, A., Gupta, S., Mathivathanan, S., Biswas, S., & Varahan, S. (2026). Glycolysis-dependent sulfur metabolism orchestrates morphological plasticity and virulence in fungi. eLife, 14, RP109075. doi: https://doi.org/10.7554/eLife.109075
Interview no 412
Explore more
🎤 Career – Real career stories and job profiles of life science professionals. Discover current opportunities for students and researchers.
💼 Jobs – The latest job openings and internship alerts across academia and industry.
🛠️ Services – Regulatory support, patent filing assistance, and career consulting services.