Reprogramming Cancer Cells to Fight Themselves: MIT Study Unveils a New Immunotherapy Strategy
Cambridge, MA: In a striking shift from conventional cancer treatment strategies, researchers have demonstrated that cancer cells themselves can be re-engineered to stimulate the immune system against tumors—turning a key weakness into a therapeutic advantage.
The study, published in Proceedings of the National Academy of Sciences (PNAS), shows that restoring a crucial immune pathway in cancer cells enables them to produce and transfer immune-activating signals, effectively helping the body recognize and attack tumors.
Hijacking Cancer Cells for Immunity
Cancer cells dominate the tumor microenvironment (TME), yet they are typically overlooked as therapeutic targets for immune activation. One major reason is the dysfunction of a critical immune signaling pathway known as cGAS–STING, which is often suppressed in tumors.
This pathway normally detects abnormal DNA within cells and triggers immune responses. However, in many cancers, the system is silenced—allowing tumors to evade immune detection.
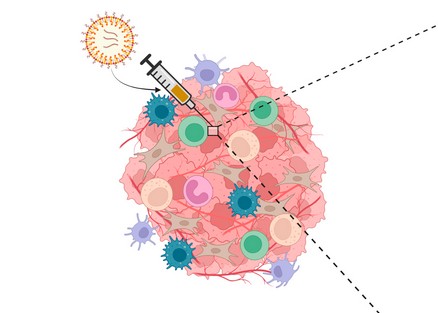
To overcome this, researchers introduced messenger RNA (mRNA) encoding cGAS, an enzyme upstream of STING, directly into cancer cells using lipid nanoparticles (LNPs). This restored the cells’ ability to produce cyclic GMP-AMP (cGAMP)—a powerful immune signaling molecule.
Turning Tumors into Signal Factories
Once reprogrammed, cancer cells began generating high levels of cGAMP. Importantly, this molecule was not confined within the cells—it was released into the surrounding environment and transferred to nearby immune cells.
This transfer activated the STING pathway in immune cells, including:
- CD8+ T cells
- Natural killer (NK) cells
- Macrophages
- Dendritic cells
The result was a coordinated antitumor immune response originating from within the tumor itself.
The approach effectively transforms cancer cells into localized factories of immune activation, bypassing the need for systemic delivery of synthetic STING agonists, which are often associated with toxicity.
Improved Tumor Control in Preclinical Models
In mouse models of melanoma, treatment with cGAS-loaded nanoparticles significantly reduced tumor growth. The therapeutic effect was further enhanced when combined with immune checkpoint blockade therapy (anti-PD-1), a widely used cancer immunotherapy.
The combination strategy not only slowed tumor progression but also amplified immune cell activation within the tumor microenvironment.
A Safer Alternative to Conventional STING Therapies
Direct administration of STING agonists has shown promise but comes with challenges, including rapid clearance from the body and systemic inflammatory side effects.
By contrast, this strategy leverages the tumor’s own cellular machinery to generate immune signals locally. This endogenous production of cGAMP reduces the need for high systemic doses and may help minimize off-target toxicity.
Additionally, because cancer cells often contain abundant cytosolic DNA—a key substrate for cGAS—the approach offers a level of inherent selectivity, preferentially activating immune responses within tumors rather than healthy tissues.
Beyond Cancer: A Platform Technology
The implications of this work extend beyond oncology. The modular nature of lipid nanoparticle delivery systems and mRNA-based programming suggests potential applications in:
- Infectious diseases
- Vaccine development
- Immune modulation therapies
By harnessing natural cellular communication pathways, the strategy represents a broader paradigm in which diseased cells can be repurposed to drive therapeutic outcomes.
Looking Ahead
While the findings are based on preclinical models, they highlight a promising direction in cancer immunotherapy—one that shifts the role of cancer cells from passive targets to active participants in their own elimination.
Future research will focus on optimizing delivery systems, understanding the precise mechanisms of cGAMP transfer, and evaluating safety and efficacy in clinical settings.
As cancer therapies evolve, the focus is increasingly on precision and adaptability. This study underscores a powerful idea: instead of fighting cancer cells from the outside, it may be possible to reprogram them from within.
If successfully translated to the clinic, this approach could redefine how the immune system is engaged in the fight against cancer—turning the disease’s own biology into its greatest vulnerability.
Source: PNAS (2025), MIT-led research
Explore more
🎤 Career – Real career stories and job profiles of life science professionals. Discover current opportunities for students and researchers.
💼 Jobs – The latest job openings and internship alerts across academia and industry.
🛠️ Services – Regulatory support, patent filing assistance, and career consulting services.