Adhesion-Controlled Arf1 Activity Coordinates Golgi Organization, Microtubule Stability, and Migration
Research Summary: Cell-matrix adhesion regulates Arf1 activity to coordinate KIF5B and dynein, maintaining Golgi organization, microtubule stability, and cargo trafficking; disruption alters Golgi architecture and impairs vesicle trafficking and cell migration.
Researcher Spotlight

Antara Chakraborty is a cell biologist with an interest in microscopy, who also enjoys cooking, baking, and travelling, and values time spent both at the bench and away from it.
Linkedin- www.linkedin.com/in/antara-chakraborty27199602
Twitter- @AntaraC9
Instagram- antarac9
Lab PI name: Prof. Nagaraj Balasubramanian, IISER, Pune
Lab social media: @AdhesionLab
What was the core problem you aimed to solve with this research?
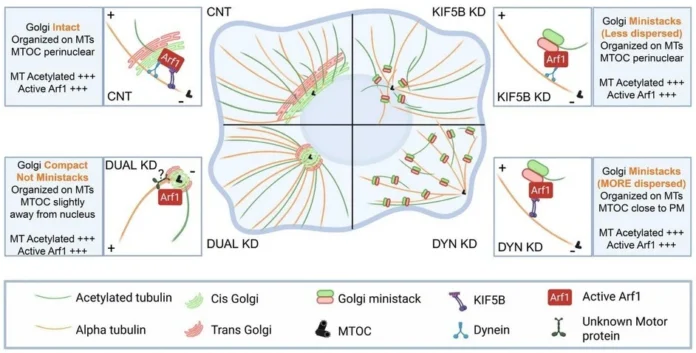
The core problem we aimed to solve was understanding how cell-matrix adhesion controls Golgi organization and function through microtubules. Specifically, we sought to uncover how adhesion-dependent changes in Arf1 activity coordinate motor proteins like KIF5B and dynein to position the Golgi and regulate microtubule stability, and how disruption of this crosstalk affects intracellular trafficking and cell behavior.
“This work highlights how understanding adhesion-dependent regulation of Golgi organization could also provide vital insights into how microtubule and motor proteins control Golgi organization and function.” – Prof. Nagaraj Balasubramanian
How did you go about solving this problem?
We addressed this by combining molecular, imaging, and functional approaches. We manipulated adhesion conditions and Arf1 activity, and used techniques such as GST-GGA3-mediated active-Arf1 pulldown, proximity ligation assays, and co-immunoprecipitation to map interactions with motor proteins. We performed siRNA-mediated knockdowns of these motor proteins to assess their roles in Golgi organization and used imaging to examine changes in Golgi architecture, microtubule organization, and MTOC positioning. Finally, we evaluated functional outcomes, including trafficking, cell spreading, polarity, and migration, to link molecular mechanisms with cellular behavior.
How would you explain your research outcomes (Key findings) to the non-scientific community?
Our research shows that a cell’s “sense of touch” with its surroundings helps maintain its internal organization. Inside every cell is a structure called the Golgi apparatus, which works like a post office, sorting and shipping important materials. We found that when cells lose their grip on their environment, this system becomes disorganized, and the internal “tracks” that help move materials also become unstable.
We discovered that a molecule called Arf1 acts as a switch, keeping this system running smoothly. It does so by coordinating tiny motor proteins that move materials to their proper destinations.
When this coordination is disturbed, the cell’s internal organization and movement are affected. This is important because such changes are often seen in diseases like cancer and neurodegenerative diseases, where cells lose control over their movement and function.
What are the potential implications of your findings for the field and society?
Our findings have important implications for both fundamental cell biology and disease understanding. We show that the Golgi is not just a passive organelle, but integrates adhesion-derived signals with motor protein activity to control its organization, vesicle trafficking, and microtubule stability. This supports an emerging view of the Golgi as responsive to physical cues such as matrix stiffness, acting through pathways involving Arf1 and its regulators.
At a mechanistic level, our work suggests that adhesion-dependent Golgi organization may directly influence microtubule modifications, opening new avenues to study how enzymes regulating tubulin post-translational modifications respond to mechanical signals.
From a broader perspective, since cytoskeletal remodeling is central to processes like cancer metastasis, immune evasion, and resistance to mechanical stress, disruption of the adhesion-Arf1-Golgi-microtubule axis could contribute to disease progression. This highlights potential targets for therapeutic intervention in cancer and other mechanobiology-related disorders.
What was the exciting moment during your research?
There were quite a few. One that stands out is the first time we observed a clear shift in the population distribution of Golgi compaction following the dual knockdown. It was both exciting and perplexing; it raised far more questions than it answered.
Another memorable moment was when we saw striking changes in the migration patterns of these cells. That result was both surprising and deeply satisfying, as it connected our molecular observations to a clear functional outcome.
Paper reference– A.Chakraborty, S. M.Pitke, R.BR, et al., KIF5B and Dynein-Dependent Golgi Organization: Role in Adhesion-Dependent Microtubule Acetylation” Traffic, no. 2 (2026): e70033, https://doi.org/10.1111/tra.70033.
Explore more
🎤 Career – Real career stories and job profiles of life science professionals. Discover current opportunities for students and researchers.
💼 Jobs – The latest job openings and internship alerts across academia and industry.
🛠️ Services – Regulatory support, patent filing assistance, and career consulting services.