Breaking the building blocks of bacterial biofilm community
Research Summary: We uncovered a previously unknown role of β2-microglobulin in selectively blocking amyloid assembly in Escherichia coli, preventing biofilm formation, and thereby introduced a host-inspired strategy to combat persistent infections.
Researcher Spotlight

Harshita Agarwal is a CSIR Senior Research Fellow who joined the PhD program in 2021 at the Functional Amyloid Biology Lab, Indian Institute of Technology Jodhpur, under the guidance of Dr. Neha Jain.
Linkedin | Twitter | Instagram
Lab: Dr. Neha Jain, Associate Professor, Department of Bioscience and Bioengineering, Indian Institute of Technology, Jodhpur
Lab Website- https://jain-lab.wixsite.com/jainlab
What was the core problem you aimed to solve with this research?
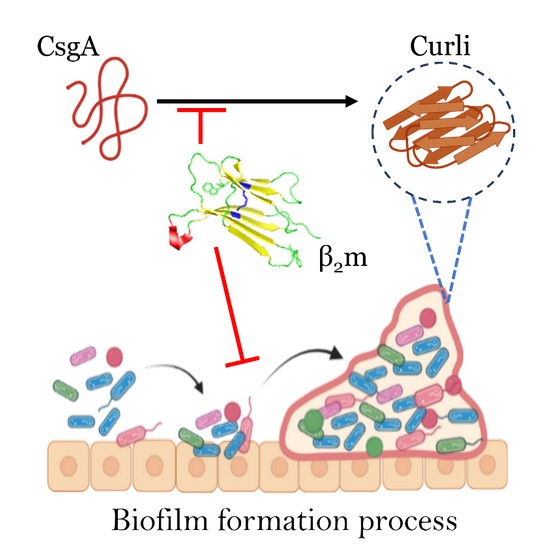
Answer: We aimed to understand how to effectively prevent bacterial biofilms at their structural foundation rather than merely killing free-floating bacteria. Biofilms formed by E. coli are highly resilient communities, protected by an extracellular matrix rich in structured protein aggregates known as amyloids. These amyloid or curli fibrils act as architectural and functional scaffolds, giving biofilms mechanical stability and remarkable resistance to antibiotics and host defenses. Despite their central role in biofilm infections, strategies to target this structural backbone remain limited. Importantly, targeting biofilm assembly rather than bacterial viability reduces selective pressure for antimicrobial resistance. We were particularly intrigued by an unexplored question: could a protein of the human immune system naturally interfere with biofilm assembly? While host proteins are well known for their antimicrobial or signaling roles, their potential involvement in targeting bacterial functional amyloids, such as curli, remains unknown. We aimed to determine whether β2-microglobulin (β2m), a human innate immune protein, could modulate curli assembly and, in turn, biofilm development. By focusing on the earliest stages of matrix formation, we targeted amyloids, the building blocks of biofilm. Ultimately, the problem was not just stopping bacterial growth, but understanding and intercepting the architectural framework that allows bacteria to build protective communities in the first place.
How did you go about solving this problem?
Answer: We began by investigating whether a naturally occurring human protein, β2m, could modulate E. coli biofilms. When we added β2m to E. coli under biofilm-forming conditions, we observed a concentration-dependent reduction in biofilm biomass. Importantly, the bacteria continued to grow normally, showing that β2m was not killing them but specifically preventing them from forming protective biofilms. Further experiments revealed that β2m acts mainly during the early stages of biofilm formation. Microscopy studies also showed that the density of the extracellular matrix of biofilm was significantly reduced in the presence of β2m.This led us to ask: how does β2m prevent biofilm formation? Biofilms in E. coli are protected by an extracellular matrix composed mainly of curli fibrils, which act as a structural scaffold supporting the bacterial community within the biofilm. We targeted curli fibrils with β2m in the lab conditions. Our Thioflavin T kinetics, turbidity assays, and electron microscopy assays showed that β2m interferes with fibril formation, preventing them from assembling into insoluble, stable structures. Finally, we tested this effect in a rat wound infection model. When β2m treatment was given to infected wounds, bacterial counts decreased, and wounds healed faster. Together, our findings show that a naturally occurring human protein can prevent bacteria from building the structural framework required for biofilm formation and could also aid in healing infected wounds.
Our study highlights the role of the host protein in reducing AMR due to biofilms by blocking biofilm matrix assembly. – Dr. Neha Jain
How would you explain your research outcomes (Key findings) to the non-scientific community?
Answer: Bacteria do not always live as single, free-floating cells. They build protective communities called biofilms that act as shields against antibiotics and the immune system. In our study, we discovered that a naturally occurring human immune protein, β2-microglobulin (β2m), can prevent E. coli from forming this protective shield in the first place. Instead of killing the bacteria, this protein prevents them from constructing curli fibrils, the structural framework that holds the biofilm together. Preventing biofilm formation at an early stage will make the bacteria more vulnerable and less capable of causing long-lasting infections. We also showed that this effect works in a wound infection model, where treatment with β2m reduced bacterial burden and promoted healing. Our research reveals that the human body already carries tools that can interfere with bacterial defence systems.
What are the potential implications of your findings for the field and society?
Answer: Our findings have important implications for both the scientific community and society at large. Our findings have direct implications for the field of biofilm biology by identifying β2m as a selective inhibitor of curli-mediated biofilm formation in E. coli. By demonstrating that β2m interferes with CsgA polymerization and prevents amyloid assembly, our study establishes curli formation as a tractable and druggable target for controlling biofilm development. This work advances the field by shifting the focus from traditional bactericidal strategies towards targeting the structural basis of biofilm formation. Most existing therapeutic approaches aim to eliminate planktonic bacteria, which often prove ineffective against biofilm-associated infections. In contrast, our study shows that selectively interfering with the assembly of matrix components can prevent biofilm establishment without affecting bacterial growth. In addition, the identification of a host-derived protein that modulates bacterial amyloids provides new insight into potential host–microbe interactions that regulate microbial community formation. From a translational perspective, our findings provide a mechanistic foundation for designing molecules that mimic the inhibitory interaction of β2m with CsgA. Overall, by uncovering a host protein that directly interferes with bacterial amyloid assembly, this work advances our understanding of biofilm regulation and provides a new conceptual and mechanistic framework for developing targeted anti-biofilm strategies.
At a societal level, preventing biofilm formation has important implications for improving public health and reducing the burden of chronic infections. Biofilm-associated infections contribute to persistent conditions. Strategies that prevent bacteria from establishing biofilms at early stages could reduce infection persistence, improve patient recovery, and lower the chances of recurrence. Insights from our study may support the development of anti-biofilm coatings for medical devices, improved wound dressings, and localized treatments designed to prevent bacterial colonization.
What was the exciting moment during your research?
Answer: Every stage of this journey was filled with learning, uncertainty, and an unusual kind of thrill. This research journey was not just about the experiments; it began with laying the foundation. The most exciting moment for me was to establish E. coli biofilm formation in the lab, which laid the foundation for this study. Further teasing apart the components of biofilms and the ability to work with them individually was equally rewarding. I could make curli amyloids in the lab, which enabled me to conduct a detailed analysis of β2m inhibition. An equally exciting moment came when we observed β2m inhibiting both biofilm formation and CsgA aggregation. Seeing this clearly during our confocal microscopy analysis and realizing that our central hypothesis was actually unfolding in the data was incredibly rewarding. Each of these moments marked an important step in my research journey.
Figure Caption: β2m interferes with curli amyloid assembly and prevents biofilm formation in E. coli
Paper reference: H. Agarwal, H. Ben, A. Chaini, B. Gurnani, N. Mukherjee, A. Pal, A.K. Upadhyaya, S. Ghosh, D. Kumar Sasmal, & N. Jain, β2-microglobulin inhibits Escherichia coli biofilm formation via selectively blocking curli assembly, Proc. Natl. Acad. Sci. U.S.A. 123 (8) e2515986123, https://doi.org/10.1073/pnas.2515986123 (2026).
Interview no 414
Explore more
🎤 Career – Real career stories and job profiles of life science professionals. Discover current opportunities for students and researchers.
💼 Jobs – The latest job openings and internship alerts across academia and industry.
🛠️ Services – Regulatory support, patent filing assistance, and career consulting services.