Sam50 Folding Mechanism Revealed: Residue-Level Insights into Mitochondrial Protein Assembly
Research Summary: Our study reveals the stepwise assembly of Sam50 with parallel folding pathways. We also identify the destabilizing hotspots that create a stability-function trade-off essential for lateral gating.
Researcher Spotlight

Roshika Ravi is a PhD graduate from the Indian Institute of Science Education and Research, Bhopal, under the guidance of Prof. R. Mahalakshmi. Her doctoral research provides a comprehensive thermodynamic and kinetic characterization of β-barrel proteins.
LinkedIn: https://www.linkedin.com/in/roshika-ravi-214376214/
Lab: Prof. R. Mahalakshmi, Indian Institute of Science Education and Research, Bhopal
Lab website: https://sites.google.com/view/prof-maha/lab-members
What was the core problem you aimed to solve with this research?
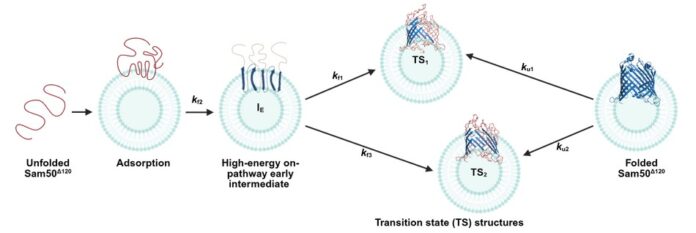
Cryo-EM structures have provided insights on how Sam50 assembles other β-barrel proteins in the outer membrane of mitochondria (OMM). However, we lack information on how it folds and the molecular factors regulating its stability, folding, and function.
How did you go about solving this problem?
A detailed thermodynamic and kinetic characterization of the transmembrane domain of Sam50 was done to understand the per-residue contribution to the stability and folding of Sam50. To deduce the magnitude of per-residue transfer free energy, we generated a library of 165 mutants by systematically substituting every transmembrane non-Ala residue to Ala and characterized their thermodynamic and kinetic properties in large unilamellar vesicles. All-atom molecular dynamics simulations and yeast survival assays were carried out to support our findings.
“We present the first mechanistic insight into how the essential mitochondrial Sam50 chaperone evolved to link folding with function and in vivo regulation.” – Prof. R. Mahalakshmi
How would you explain your research outcomes (Key findings) to the non-scientific community?
Cells rely on many essential proteins, one of which is Sam50, located in the outer membrane of mitochondria (OMM). It helps in the assembly of β-barrel proteins in the OMM. For Sam50 to work, it must fold correctly from a thread-like chain into a defined barrel structure. Here we show the two major folding pathways followed by Sam50 and reveal how its building block (amino acids) contributes to reaching the end state structure and its function.
What are the potential implications of your findings for the field and society?
Our study bridges Sam50–dependent mechanisms of β-barrel folding with microscopic regulatory elements vital for the assembly and function of the core Sam50 β-barrel itself. Elucidation of early intermediates in Sam50 folding and correlation with in vivo function will provide mechanistic details of mitochondrial biogenesis at the atomic level.
What was the exciting moment during your research?
The entire process, from performing the experiments to interpreting the data, was incredibly exciting. But what I found most satisfying was bridging those experimental observations with their biological roles. For example, linking the destabilized hotspot to its functional role in lateral gating and how the non-native interactions formed in the C-terminus explain why Sam50 needs assistance from SAM complex for its folding.
Paper reference/citation (with link): Ravi, R., Gupta, S., Kumari, J. et al. Linking multipoint folding and stability with functional regulation in the mitochondrial transmembrane β-barrel Sam50. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70904-3
Explore more
🎤 Career – Real career stories and job profiles of life science professionals. Discover current opportunities for students and researchers.
💼 Jobs – The latest job openings and internship alerts across academia and industry.
🛠️ Services – Regulatory support, patent filing assistance, and career consulting services.