New Breakthrough in Myotonic Dystrophy 1: Myc Identified as Potential Therapeutic Target
Research Summary: Our research shows that Myc is a key player in Myotonic Dystrophy 1. We observed that Myc could reduce the disease progression and reduce RNA toxicity in the DM1 model.
Researcher Spotlight
Dipti Chakraborty is a PhD scholar in the MDS Laboratory at the National Brain Research Centre, India. Her research focuses on Myotonic Dystrophy 1 using Drosophila melanogaster to understand molecular mechanisms.
LinkedIn: linkedin.com/in/dipti-chakraborty-6035a7401
Twitter: https://x.com/DIPTICH80762410
Instagram: https://www.instagram.com/dipti__chakraborty
Lab: Mayanglambam Dhruba Singh, BRIC-National Brain Research Centre
Lab website: https://www.nbrc.ac.in/newweb/research/groups/mdhruba
What was the core problem you aimed to solve with this research?
The study aimed to address the lack of clearly defined molecular mechanisms and key regulatory factors in Myotonic Dystrophy Type 1 (DM1), a complex genetic disorder driven by toxic CTG repeat expansions. Specifically, our goal was to identify critical molecular players that contribute to disease progression and could be targeted to reduce cellular toxicity and improve disease outcomes.
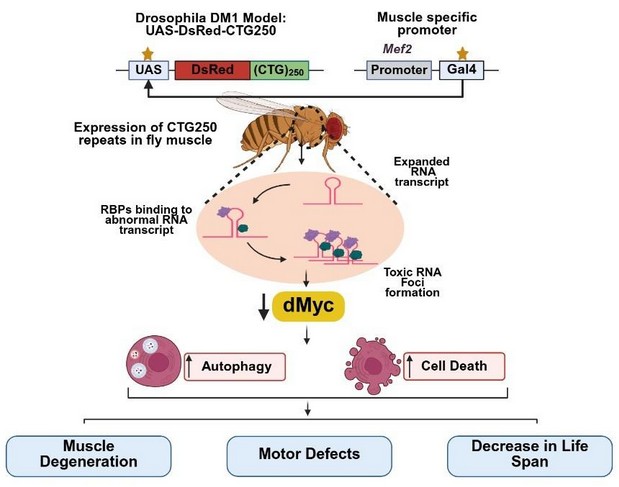
How did you go about solving this problem?
We approached this problem using a well-established Drosophila model of DM1 that expresses expanded CTG repeats to mimic key disease features. To investigate disease, we employed a fly model expressing pathogenic CTG repeat expansions (CTG250 and CTG270), which successfully recapitulates key features of DM1, including RNA foci formation, muscle fibre degeneration, impaired locomotion, and reduced lifespan. Using this model, we identified dMyc, a conserved homolog of human Myc, as a novel genetic modifier of CTG-induced toxicity. We modulated dMyc expression and assessed its effects through phenotypic, molecular, and cellular assays. To evaluate the impact, we combined multiple approaches, including phenotypic assays (eye morphology and locomotion), lifespan, molecular and cellular analyses, and gene/protein expression studies. We then performed genetic manipulations to modulate dMyc and related pathway expression.
This integrated strategy enabled us to systematically assess how altering dMyc levels affects disease progression and underlying cellular mechanisms, including autophagy and cell death.
“This study provides interesting insights into Myotonic Dystrophy 1 pathogenesis and highlights a potential strategy to halt disease progression.” – Mayanglambam Dhruba Singh
How would you explain your research outcomes (Key findings) to the non-scientific community?
Myotonic Dystrophy 1 is a muscular disease. However, DM1 has recently been identified as a multisystemic disease affecting the brain, heart, and muscles, among other organs. No drug is available for this disease so far. This study aims to find mechanisms that could be utilised to treat this deleterious disease. This study has identified a biological mechanism that could be targeted to suppress the idiopathic progression of DM1. Therefore, our findings provide interesting insights into the mechanism of DM1 disease and potential therapeutic applications.
What are the potential implications of your findings for the field and society?
From a research perspective, this work identifies dMyc as a novel and critical regulator in DM1. It opens a new direction for understanding how cellular processes such as autophagy and cell death contribute to disease progression and highlights Myc modulation as a promising therapeutic strategy. This can guide future studies toward the development of targeted treatments and the exploration of similar mechanisms in other neurodegenerative or muscular disorders.
From a societal perspective, DM1 currently has limited treatment options. These findings offer hope for improved therapeutic approaches that could enhance muscle function, slow disease progression, and improve patients’ quality of life. In the long term, such research helps reduce the healthcare burden and improve patient care for individuals affected by genetic disorders such as DM1.
What was the exciting moment during your research?
The most exciting moment in our research was when we first observed a clear phenotypic rescue in the fly, an initial and encouraging indication that our approach was working. This early result motivated us to explore the underlying mechanisms further.
As the study progressed, the observation of significant improvements in muscle structure, movement, and lifespan following restoration of dMyc in the DM1 model was a true turning point. It marked the transition from simply observing phenotypic rescue to understanding its molecular basis.
Additionally, working with Drosophila muscle tissue is technically challenging, and during this study, we have standardised many techniques. Successfully standardising these experiments and achieving consistent results heightened the excitement and significance of the project.
Paper reference: Chakraborty, D., Singh, N.T., Borthakur, S. et al. dMyc suppresses CTG-induced cytotoxicity in the Drosophila model of DM1 by reducing autophagy and cell death. Cell Death Discov. (2026). https://doi.org/10.1038/s41420-026-03123-w (https://www.nature.com/articles/s41420-026-03123-w#citeas) (*These authors contributed equally)
New Book Launched – Molecules, Mentors & Mindsets: Building Indian Biopharma
Buy your copy today: https://biopatrika.com/science-society/book-molecules-mentors-mindsets-building-indian-biopharma-biocon/
Book Launch: Molecules, Mentors & Mindsets: Building Indian Biopharma | Biocon Focus
Online Interactive Course on Immunology | Starts form June 2026 | Register Today
Online Interactive Course on Immunology | Starts form June 2026 | Register Today




