Unlocking the Secrets of Akkermansia muciniphila: How a Gut Specialist Navigates the Sulfated Mucin Barrier
Akkermansia muciniphila (A. muc), an obligate mucin degrader has earned its reputation as a “sentinel” of the human gut. This bacteria lives within the protective mucus layer of the gut and makes up about 3% of the total human colonic microbiota (HCM). Its presence is frequently associated with positive health outcomes like reduced inflammation and improved metabolic health. However, the precise molecular mechanisms adopted by Akkermansia to thrive in the harsh colonic environment has been relatively underexplored until recently.
A study by Dey et al., in bioRxiv, provides a comprehensive look at the “sulfobiology” of A. muc, revealing how its unique suite of carbohydrate sulfatases allows it to utilize colonic mucin in ways that distinguish it from other gut bacteria like Bacteroides.
The Mucin Barrier and the Role of Sulfation
Colonic mucin is made up of MUC2 which structurally is a massive, gel-forming glycoprotein that serves as both a physical barrier and a primary nutrient source for mucin lovers like A. muc. Up to 80% of MUC2 consists of complex O-glycans. Importantly, these glycans are heavily sulfated, particularly in the distal regions of the colon, where sulfation can reach up to 10% by mass. To access it mucinophilic gut bacteria produce enzymes called carbohydrate sulfatases that can remove these sulfation. While excessive degradation of this mucus layer by some bacteria is linked to inflammatory bowel diseases (IBD) like ulcerative colitis, A. muc is generally viewed as a beneficial member of the community. Understanding its sulfatase toolkit is therefore essential for understanding its beneficial effects.
A Preference for Glycopeptides
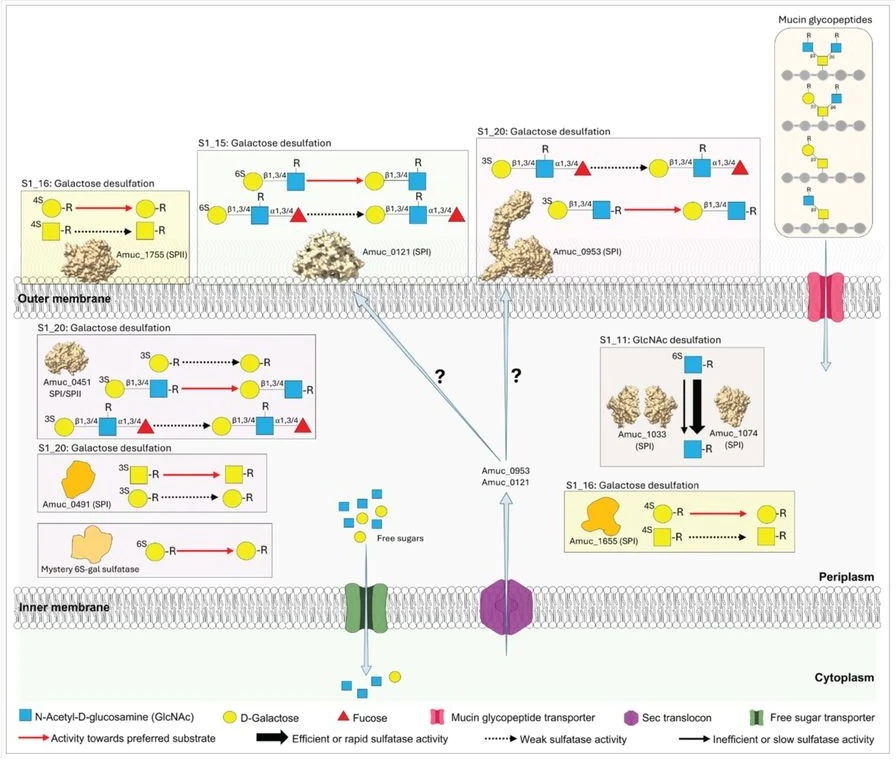
One of the study’s most interesting findings is A. muc requires glycopeptides/protein forms of colonic mucin for metabolism and its sulfatases have unique adaptations compared to Bacteroides species. The researchers provide kinetic information for 8 of the 10 A. muc S1 sulfatases, define their cellular location, and structurally characterize 5 of these enzymes. These enzymes are finely tuned to remove sulfate groups from specific positions on different sugars:
- S1_11 Sulfatases (Amuc_1033 & Amuc_1074): These enzymes target 6-O-sulfated N-acetyl-D-glucosamine (6S-GlcNAc). Interestingly, this activity is restricted exclusively to the periplasm, the space between the inner and outer bacterial membranes.
- S1_16 Sulfatases (Amuc_1655 & Amuc_1755): Unlike their Bacteroides counterparts, which act on both Galactose and N-acetyl-D-galactosamine (GalNAc), these A. muc enzymes show a strong specificity for 4-O-sulfated D-galactose (4S-Gal).
- S1_15 Sulfatase (Amuc_0121): This enzyme is located on the cell surface and is capable of desulfating 6S-Gal and 6S-GalNAc, including complex Lewis structures found on the ends of mucin chains.
- S1_20 Sulfatases (Amuc_0451, Amuc_0491, & Amuc_0953): These are large, multi-domain enzymes that target 3-O-sulfated sugars like 3S-Gal and 3S-LacNAc.
Discovery of a Novel Mucin Binding Domain
Perhaps the most significant structural discovery is a “new family of mucin-binding domain” attached to the N-terminus of Amuc_0953 sulfatase. This domain features a large parallel β-helix domain, specifically to bind to colonic mucin. Intriguingly, this module is almost entirely restricted to the Akkermansia genus, suggesting it is a unique adaptation that possibly allows these A. muc to anchor themselves to the colonic mucus.
Clinical Implications: Precision Gut Health
The distinct differences in how A. muc and Bacteroides species process sulfated mucins offer a new path for therapeutic intervention. Because Bacteroides sulfatases are known to drive disease in susceptible models of IBD, these enzymes are potential drug targets. The unique structural adaptations identified in A. muc sulfatases could allow for the development of drugs that inhibit harmful Bacteroides sulfatases while sparing the beneficial Akkermansia. By mapping the molecular landscape of A. muc’s sulfatases, this research provides a vital blueprint for understanding how one of our most important gut residents maintains its niche and contributes to a healthy intestinal barrier.
Preprint link: https://www.biorxiv.org/content/10.1101/2025.09.11.675649v1
Explore more
🎤 Career – Real career stories and job profiles of life science professionals. Discover current opportunities for students and researchers.
💼 Jobs – The latest job openings and internship alerts across academia and industry.
🛠️ Services – Regulatory support, patent filing assistance, and career consulting services.



