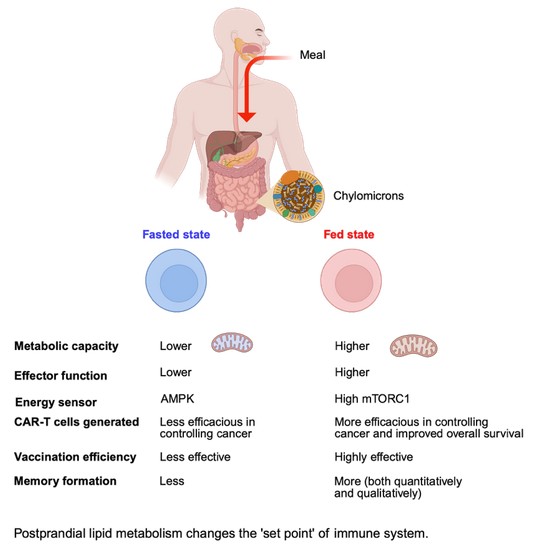
Postprandial Lipid Metabolism Reprograms T Cells
Research Summary: T cells rapidly sense metabolic changes after eating. Postprandial metabolites, especially diet-derived lipids carried by chylomicrons, enhance T cell metabolism, mitochondrial function, translation, proliferation, and effector activity through the ‘Chylomicron-LDLR-mTOR signaling axis’. These effects persist long-term and improve CAR-T cell performance, showing that even brief exposure to post-meal metabolites can significantly shape immune responses and therapeutic potential.
Researcher Spotlight
Dr. Alok Kumar, an Indian national and IIT Roorkee alumnus, completed his doctoral studies from Kyoto University, Kyoto, Japan (under Prof. Tasuku Honjo (Nobel Prize, Medicine, 2018)) and moved to the United States for his postdoctoral research work under Prof. Greg M Delgoffe at the University of Pittsburgh.
LinkedIn: https://www.linkedin.com/in/kumaralus21np40/
Twitter/X: https://x.com/DrAlok_np40
Lab: Dr. Greg M Delgoffe, Professor, University of Pittsburgh, Department of Immunology, Pittsburgh, PA, USA.
Lab social media: @DelgoffeLab
What was the core problem you aimed to solve with this research?
Ans. While scientific literature has established that cell-intrinsic metabolic pathways and local environments regulate immune function, the impact of acute dietary inputs remains poorly studied. Our research aims to address the significant gap in our understanding of how systemic metabolic flux, specifically the short-term changes that occur after a meal (postprandial state), influences T cell immunity. Our study examines how T cells perceive these immediate systemic changes and how such perception influences their metabolic and functional set points. We sought to determine if the timing of a meal relative to T cell activation or cell harvest can fundamentally reprogram immune responses and improve the efficacy of clinical interventions like vaccinations and cellular therapies to treat cancer and other immune-associated malfunctions.
How did you go about solving this problem?
Ans. To address this issue, we first collected PBMCs from humans after an overnight fast and again 6 hours post breakfast/lunch. We found that immune T cells are acutely sensitive to systemic changes in metabolism, such that T cells harvested after a meal are metabolically and functionally superior to those isolated in the fasted state. To our surprise, the T cells from the fed status maintained their high metabolic and effector functions even after long expansion in vitro. To decipher the mechanism, we performed transcriptomics, epigenetic study, and proteomics and found that T cells in a fed state exhibit heightened mTORC1 (a key cellular sensor that is activated when energy and nutrients are plentiful), which results in increased protein translation of metabolic and immune genes.
“Eating a meal changes the ‘set point’ of the immune system and those T cells are now primed to activate protein translation and take off when they are stimulated to fight infections or kill cancer cells.” – Dr. Greg M Delgoffe
How would you explain your research outcomes (Key findings) to the non-scientific community?
Ans. When our grandmothers used to say a very popular adage: ‘starve a fever, feed a cold’. I was wondering about the scientific basis of these statements, but now I think there’s some value in this. Previous research suggests the effect of long-term intake of different kinds of diets on immune responses, while how the nutrient-deficient (fasted status) and nutrient-proficient (fed or postprandial status) which happens to us in our daily routine has any impact on immune responses is not well studied. Inspired by the popular saying ‘feed a cold’, we asked if the immune cells behave differently if they come from a fasted or fed host. We found that the T cells from the same human donor collected post-meal had higher metabolic capacity and effector functions compared to T cells collected in an overnight fasted condition from the same donor. Our findings have great clinical implications in a) cancer treatment (CAR-T cell-based adoptive cell therapy b) vaccination, and c) immune analysis. In very simple words, if a person has been jabbed post-meal, they will have a better immune response compared to the other person who was jabbed in the fasted state.
What are the potential implications of your findings for the field and society?
Ans. Our research indicates that the immune system is exquisitely sensitive to immediate dietary changes. It means that the nutritional state of an individual at the time of T cell activation or priming has a profound impact on their long-term immune response. This discovery has several major implications and benefits for human health.
- CAR-T cells manufactured from PBMCs collected after a meal are functionally superior and provide a therapeutic advantage compared to those harvested in a fasted state.
- Vaccination outcomes may be influenced by whether an individual is fed or in a fasting state. If individuals receive vaccines in a fasted state, the resulting immune memory and protection might be weaker than if they were vaccinated after eating.
- Currently, most clinical analyses of human immunity do not account for the nutrient status of the donor at the time of blood or tissue collection. Our study suggests that a significant portion of the unexplained variability in human immune studies may be attributed to postprandial metabolism, underscoring the need to standardize nutritional status during medical testing to achieve more accurate results. This highlights a massive, previously overlooked factor in medical research and vaccine efficacy.
What was the exciting moment during your research?
Ans. There were two exciting moments during this research, if I remember correctly. The first moment was when we found by comparison of transcriptomics and proteins, that it is the mTORC1 signaling that is responsible for postprandial metabolic benefit in fed status over fasted status. To confirm this, we inhibited mTORC1 in vivo while mice were fed ad libitum. We found that mTORC1 inhibition cancels the postprandial benefit of fed status, as evidenced by mTORC1-inhibited fed OT-I T cells being less effective in clearing viral load in Vaccinia-OVA infection co-transfer model. This was the moment of excitement.
Further, for the clinical aspect of this finding, we generated hCD19 CAR-T cells from T cells collected from the same human donor either in fasted or fed status and tested their efficacy in NALM6 lymphoma-bearing NSG mice. We found that CAR-T cells generated from fed status were far superior in eradicating lymphoma and improving the survival of the host. This was an exciting moment for the team, as our findings are not just a mere discovery in basic biology, but rather, they have the potential to improve cancer treatment outcomes.
Paper reference: https://www.nature.com/articles/s41586-026-10432-8
New Book Launched – Molecules, Mentors & Mindsets: Building Indian Biopharma
Buy your copy today: https://biopatrika.com/science-society/book-molecules-mentors-mindsets-building-indian-biopharma-biocon/
Book Launch: Molecules, Mentors & Mindsets: Building Indian Biopharma | Biocon Focus
Looking for career opportunities. Visit jobs.biopatrika.com