Synthesis of Novel Cyclopropylallocolchicinoids via Interrupted Corey-Chaykovsky Reaction: A New Class of Potent Antimitotic Agents.
Research Summary: We have developed a new class of sp3-rich, cyclopropyl-fused allocolchicinoids by tapping into the unique nucleophilic character of Corey-Chaykovsky ylides. Beyond their novel structure, these molecules have proven to be potent microtubule disruptors and promising blockers to Influenza A virus infections.
Researcher Spotlight
Dipto Mukhopadhyay is a PhD student at IISER Mohali, specialising in developing new methodology in organic chemistry through phosphine catalysis or sulfur ylides.
Linkedin: https://www.linkedin.com/in/dipto-mukhopadhyay-21746b192
Twitter: https://x.com/DiptoMukhopadhy
Lab: Prof. S. S. V. Ramasastry, Indian Institute of Science Education and Research (IISER), Mohali
Linkdin: https://www.linkedin.com/in/s-s-v-ramasastry-29190945
Twitter: https://x.com/ramsastrys
What was the core problem you aimed to solve with this research?
The colchicine family is well-known for its role in treating gout, relaxing muscles, and stopping cell division. Within this group, molecules like colchicine, allocolchicine, and cyclopropyl allocolchicinoid (CAC) are particularly explored and investigated for their anti-mitotic properties. However, making them in a lab is challenging. Current methods are often too long, and low-yielding, or rely on ‘tricky’ chemistry that isn’t sustainable. These roadblocks make it hard to study how their structures relate to their activity, which is exactly what we need to do to find more potent drug candidates.
“We developed short and efficient synthesis of cyclopropylallocolchicinoids that disrupt microtubule organization, leading to potent antimitotic and anti-influenza activity.” – Prof. S. S. V. Ramasastry
How did you go about solving this problem?
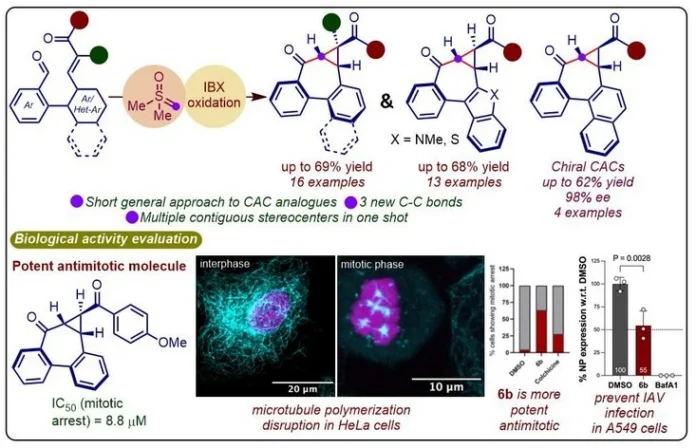
We’ve recently moved beyond the typical use of the Corey-Chaykovsky reagent for simple epoxidation or cyclopropanation. Our group has developed a new class of one-pot cascade reactions that take a different path. By using dimethyloxosulfonium methylide (DOSM), we’ve been able to build several unique cyclopropyl-tethered structures in a single step, showing just how versatile this chemistry can be. In this communication, we have shown the first general approach to the synthesis of sp3-rich dibenzocycloheptanes using DOSM from our well-designed substrates. We’ve successfully synthesized a diverse collection of arene and heteroarene-fused CACs. What’s particularly exciting is that the majority of these structures have never been described in the literature until now.
How would you explain your research outcomes (Key findings) to the non-scientific community?
While synthesizing new organic molecules is often seen as an art, the true challenge lies in creating structures that can meaningfully interact with complex biological systems. Cell division is a masterclass in synchronization, and when it falters, the result is often tumour growth. We have developed a new class of molecules designed to step into this complex environment and successfully halt uncontrolled division. We believe this study opens a new door toward a family of biologically significant candidates with real therapeutic potential.
What are the potential implications of your findings for the field and society?
Beyond the synthetic novelty, our CAC scaffold serves as a versatile platform for creating intricate molecular architectures. Given their structural kinship with allocolchicinoids, we anticipated they would interact strongly with microtubules, the cellular ‘highways’ essential for everything from cell division, intracellular transport to viral transport. High-content imaging confirmed this, revealing that molecule 6b induces mitotic arrest by disrupting microtubule dynamics. Additionally, we found that 6b effectively blocks Influenza A (IAV) infection in human lung epithelial cells by perturbing the very microtubule and centrosome organization the virus relies on to thrive
What was the exciting moment during your research?
I first encountered colchicine in a school classroom, where my teacher explained its basic ability to bind with microtubules. At the time, it was just an introductory concept in a textbook. Seeing our own molecule outperform that classic standard years later was a true ‘full-circle’ moment for us-it was easily the most exciting breakthrough of this entire project
Paper reference/citation (with link): Sulfur Ylide-Mediated Cascade Transformations to Access Cyclopropylallocolchicinoids with Potent Antimitotic Activity. Mukhopadhyay, D.; Patel, K.;#Chattopadhyay, A.;#Pal, S.;#Pathak, T.; Banerjee, I.; Ramasastry, S. S. V. JACS Au. 2026, 10.1021/jacsau.6c00268 [#Contributed equally to the work]
https://doi.org/10.1021/jacsau.6c00268
New Book Launched – Molecules, Mentors & Mindsets: Building Indian Biopharma
Buy your copy today: https://biopatrika.com/science-society/book-molecules-mentors-mindsets-building-indian-biopharma-biocon/
Book Launch: Molecules, Mentors & Mindsets: Building Indian Biopharma | Biocon Focus
Online Interactive Course on Immunology | Starts form June 2026 | Register Today
Online Interactive Course on Immunology | Starts form June 2026 | Register Today