New Study Reveals the Lipid Paradox Driving Fatty Liver Disease Progression in MAFLD
Research Summary: We identified a paradoxical hepatic “switch” where triglyceride synthesis protects against lipotoxicity and fibrosis, yet simultaneously drives lipid accumulation, thereby delaying severe damage while promoting early MAFLD progression.
Researcher Spotlight
Atanu Mondal is currently working as a Senior Research Fellow at the Saha Institute of Nuclear Physics, Kolkata. His research focuses on understanding the epigenetic regulation of metabolic pathways, contributing to disease development.
LinkedIn: https://www.linkedin.com/in/atanu-mondal-255470325
Twitter: https://x.com/Atanu_Mondal96
Instagram: https://www.instagram.com/willnever_walk_alone
Lab: Prof Chandrima Das, Saha Institute of Nuclear Physics, Kolkata
Lab social media: https://www.linkedin.com/in/chromatin-dynamics-lab-500b39277
Lab Website: https://chandrimadascdl.wixsite.com/cromatindynamicslab
What was the core problem you aimed to solve with this research?
Contemporary lifestyle patterns, particularly calorie-dense diets and reduced physical activity, have led to a sharp rise in metabolic disorders, among which Metabolic dysfunction-associated fatty liver disease (MAFLD) has emerged as a major global concern. It is estimated to affect nearly 40% of the global population, underscoring its significant public health burden. MAFLD is a multifactorial condition driven by obesity, insulin resistance, Type 2 diabetes (T2D), dyslipidemia, and excessive intake of saturated fatty acids, all of which collectively disrupt hepatic lipid homeostasis and metabolic balance.
The disease progresses along a continuum, beginning with simple steatosis, referred to as Metabolic dysfunction-associated steatosis (MAS), and advancing to Metabolic dysfunction-associated steatohepatitis (MASH), which is characterized by hepatocellular injury, inflammation, and varying degrees of fibrosis. If left unchecked, this progression can culminate in cirrhosis and ultimately Hepatocellular carcinoma. Despite extensive research, the molecular mechanisms that govern the transition from benign lipid accumulation to inflammatory and fibrotic stages remain incompletely understood.
At early stages, the liver adapts to lipid overload by increasing triglyceride storage, leading to hepatomegaly. Importantly, this stage remains reversible through lifestyle interventions such as dietary modification and physical activity. However, as the disease advances, persistent lipotoxic stress triggers inflammatory signalling and fibrogenic responses, making the condition progressively less reversible and more clinically severe. Current therapeutic approaches are largely centred on reducing hepatic triglyceride accumulation. While this strategy addresses early lipid overload, it does not adequately target the downstream mechanisms that drive inflammation and fibrosis.
This highlights a critical research gap: the need to understand the molecular pathways that regulate the shift from adaptive lipid storage to pathological inflammation and fibrogenesis. In the present study, we aim to address this gap by focusing on the mechanisms underlying disease progression, particularly the transition to MASH. By targeting these pathways, we propose that halting or slowing the progression at this stage could preserve liver function and enhance the effectiveness of lifestyle-based interventions, ultimately improving disease outcomes.
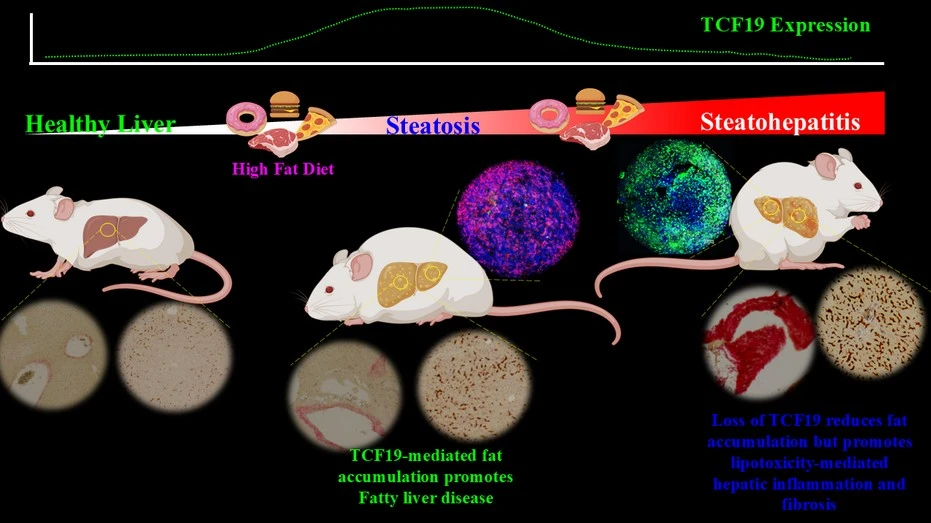
How did you go about solving this problem?
To address this problem, we established a diet-induced MAS and MASH mouse model that recapitulates key pathological features of metabolic liver disease. We employed an integrative multi-omics approach to systematically connect epigenetic alterations with dynamic changes in hepatocellular metabolic processes. By combining transcriptomic, proteomic, and metabolomic analyses, we were able to generate a comprehensive molecular landscape of disease progression. Through this integrated analysis, we identified the epigenetic reader protein TCF19 as a critical regulator that transcriptionally governs the expression of multiple fatty acid elongase genes, primarily by modulating the chromatin state at their promoter regions and thereby influencing gene accessibility and activity. We identified a novel histone modification dynamic regulated by the TCF19-associated coregulatory complex: histone H3K4 trimethylation (H3K4me3)-dependent changes of CBP/p300-mediated histone H3K27 acetylation (H3K27ac), which leads to the activation of elongase genes. In addition, we investigated the interplay between hepatocytes and components of the immune system, with a particular focus on understanding how this cellular crosstalk contributes to the onset and progression of hepatic inflammation and fibrosis, ultimately providing mechanistic insight into disease pathogenesis.
“The liver’s protective lipid buffering can paradoxically drive early fatty liver disease through adaptive metabolic reprogramming” – Prof Chandrima Das
How would you explain your research outcomes (Key findings) to the non-scientific community?
Our study provides insight into how the liver’s intrinsic defence mechanisms respond to metabolic stress by actively promoting lipid accumulation as a protective strategy against severe forms of MAFLD. These findings highlight a fundamental paradox in liver biology: a mechanism that is intended to preserve cellular integrity can, over time, contribute to disease initiation. In this context, we identified the transcription factor TCF19 as a central metabolic gatekeeper that orchestrates this response. Under conditions of lipotoxic stress, TCF19 facilitates the conversion of harmful lipid intermediates into triglycerides, which are comparatively inert and can be safely stored within lipid droplets. This adaptive response reduces cellular toxicity, thereby protecting hepatocytes from damage and delaying progression toward more advanced pathological states such as inflammation and fibrosis. At the same time, however, this protective mechanism inadvertently promotes the accumulation of lipids within the liver, contributing to the onset of early-stage fatty liver disease.
Thus, while TCF19-mediated metabolic reprogramming serves as an immediate safeguard against lipotoxic injury, it also creates a permissive environment for lipid buildup, ultimately linking cellular protection with disease initiation. This dual role underscores the intricate balance and complexity of metabolic regulation in the liver, where protective and pathological processes are tightly intertwined.
What are the potential implications of your findings for the field and society?
Our findings call into question long-standing therapeutic strategies that focus predominantly on reducing hepatic fat content as the primary intervention. Instead, our study emphasizes that a more effective and sustainable approach may involve fine-tuning the way lipids are processed, compartmentalized, and balanced within hepatocytes. By shifting the therapeutic perspective from mere fat reduction to a deeper understanding of metabolic regulation and lipid handling, our work opens new avenues for the development of targeted, mechanism-driven, and more nuanced interventions.
In the context of the rapidly increasing prevalence of MAFLD, particularly among urban populations in India, where lifestyle transitions are driving metabolic disorders, our study provides timely and relevant insights. Beyond advancing the fundamental understanding of liver biology, these findings establish a conceptual framework for designing more precise and context-specific therapeutic strategies. Ultimately, this work contributes to addressing a growing public health challenge by guiding the development of interventions that are better aligned with the underlying biology of the disease.
What was the exciting moment during your research?
One of the most exciting moments in this journey came during a late-night realization that triglyceride production, contrary to our initial assumption, was playing a hepatoprotective rather than a hepatotoxic role. This unexpected insight fundamentally shifted the direction of our study and guided us toward resolving a previously unaddressed aspect of the problem. The process was further enriched by numerous intense discussions and debates with Prof. Chandrima Das, which continuously challenged our interpretations and shaped the study in an intellectually stimulating way. After months of repeated failures and experimental setbacks, the moment we finally obtained consistent and meaningful results was immensely rewarding.
I also vividly remember my first meeting and in-depth discussion with Prof. Siddhesh S. Kamat, which significantly influenced my understanding of lipid metabolic pathways, particularly the mechanisms underlying fatty acid chain elongation. In addition, the time spent working at IISER Pune with Dr. Arnab Chakraborty remains especially memorable, as it provided both valuable scientific exposure and enriching collaborative experiences.
Paper reference: Mondal, A., Chakraborty, A., Nandi, S., Singh, V., Kamat, S. S., and Das, C. Transcription factor 19 modulates fatty acid elongation and unfolded protein response to attenuate palmitic acid-induced hepatic dysfunction. Nat Commun (2026). https://doi.org/10.1038/s41467-026-72138-9
New Book Launched – Molecules, Mentors & Mindsets: Building Indian Biopharma
Buy your copy today: https://biopatrika.com/science-society/book-molecules-mentors-mindsets-building-indian-biopharma-biocon/
Book Launch: Molecules, Mentors & Mindsets: Building Indian Biopharma | Biocon Focus





