Human Gut Cells Use Quorum-Sensing Mimics to Communicate with Microbiome
What if human gut cells could communicate with microbes using the same chemical signals bacteria use among themselves? New research suggests this may indeed be happening—revealing a previously hidden layer of communication between host cells and the gut microbiome.
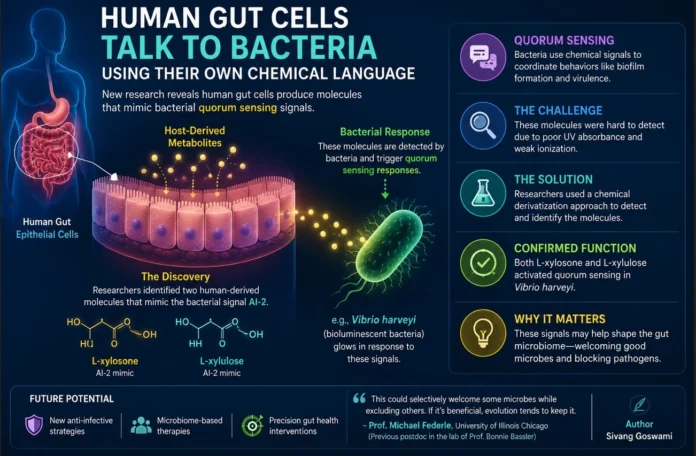
Scientists have identified and characterized metabolites produced by human gut epithelial cells that mimic bacterial quorum-sensing molecules. Quorum sensing is a well-known bacterial communication system that allows microbes to coordinate behaviors such as biofilm formation and virulence—key processes for survival and colonization.
Earlier studies had shown that eukaryotic organisms, including yeast, can produce mimics of autoinducer-2 (AI-2), a universal bacterial signaling molecule. These mimics arise as metabolic byproducts and can interact with bacterial receptors. Human gut cells were also known to generate AI-2–like activity, but the identity of these molecules had remained elusive.
A major challenge in identifying these compounds has been technical. These molecules lack strong ultraviolet (UV) absorbance and ionize poorly, making them difficult to detect using conventional analytical approaches.
To overcome this, researchers in the lab of Prof. Bonnie Bassler (Princeton University) developed a chemical derivatization strategy. By reacting metabolites from nutrient-deprived gut epithelial cells with 1,2-diamino-4,5-methylenedioxybenzene (DMB), they converted them into quinoxaline derivatives that can be readily detected using UV spectroscopy and identified through mass spectrometry.
This approach led to the discovery of L-xylosone, a compound structurally similar to bacterial AI-2. Functional validation using a reporter strain of Vibrio harveyi—which emits light in response to quorum-sensing signals—confirmed that L-xylosone can activate bacterial communication pathways.
Further investigation revealed that L-xylulose, a related pentose sugar, also triggers similar responses. While L-xylulose is known to arise as a metabolic intermediate, the pathways leading to L-xylosone production in mammalian systems remain poorly understood.
For Prof. Michael Federle of the University of Illinois Chicago—who previously trained with Bonnie Bassler—these findings point to intriguing biological possibilities.
“For residing microorganisms or incoming species, this could be beneficial—or it could selectively welcome certain microbes while excluding others,” he said. “If such interactions confer an advantage, they may be retained through evolution—but demonstrating that experimentally is the real challenge.”
Beyond fundamental biology, the findings hint at potential therapeutic applications. If these host-derived molecules can shape microbial communities—by promoting beneficial microbes or suppressing pathogens—they could become targets for future microbiome-based interventions.
“If these metabolites help prevent pathogen colonization—perhaps by enabling beneficial microbes to produce antimicrobial factors—that could be advantageous,” Dr. Federle added. “But understanding this will require integrating metabolite profiling with metagenomic analyses to track how microbial populations respond.”
The study opens a new frontier in microbiome research, suggesting that human cells are not passive hosts but active participants in microbial communication. Future work will focus on uncovering the metabolic origins of these molecules and their precise role in shaping gut health and disease.

Reference study: Discovery of a Human Metabolite That Mimics the Bacterial Quorum-Sensing Autoinducer AI-2. JACS 2026. https://pubs.acs.org/doi/10.1021/jacs.5c18527
About the Author:
Sivang Goswami is a PhD student at IISER Pune studying the fundamental mechanisms of bacterial division in cell wall-less bacteria. He completed his undergraduate degree in Microbiology from the University of Delhi and a Master’s in Biotechnology from South Asian University. Passionate about science communication, Sivang enjoys writing stories and poetry to make fascinating scientific ideas accessible to general audiences.
Social Media:
Instagram: https://www.instagram.com/itisshivang
LinkedIn: https://www.linkedin.com/in/sivang-goswami-a31318175
New Book Launched – Molecules, Mentors & Mindsets: Building Indian Biopharma
Buy your copy today: https://biopatrika.com/science-society/book-molecules-mentors-mindsets-building-indian-biopharma-biocon/
Book Launch: Molecules, Mentors & Mindsets: Building Indian Biopharma | Biocon Focus